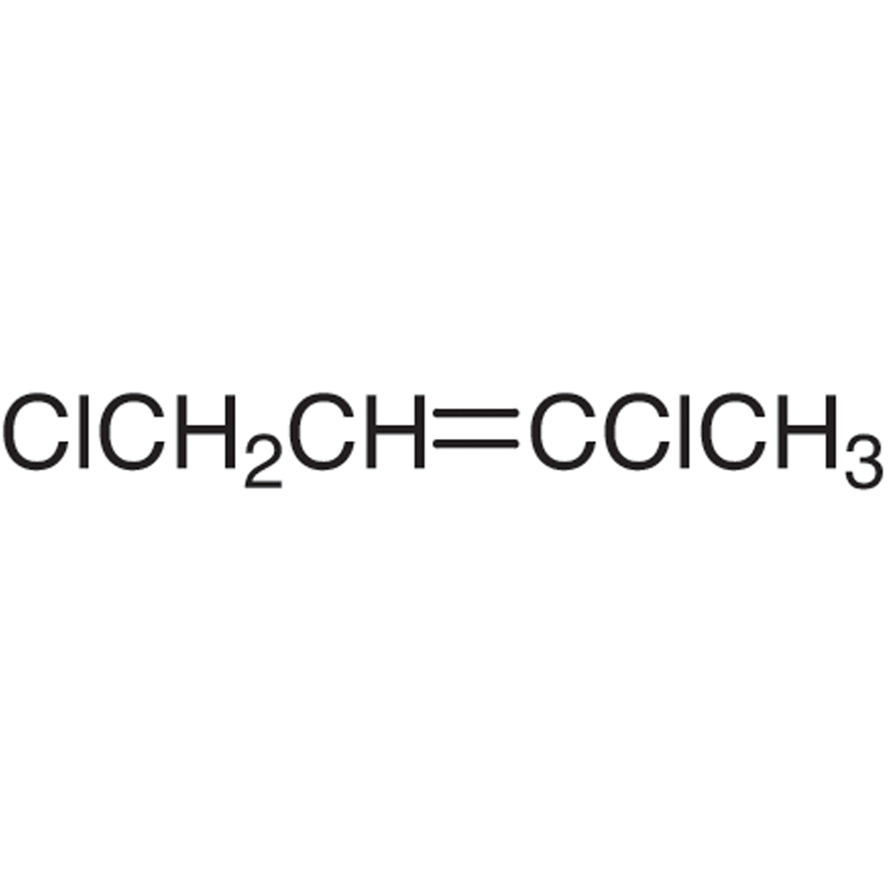
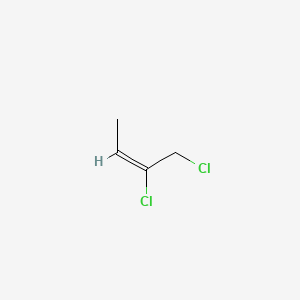
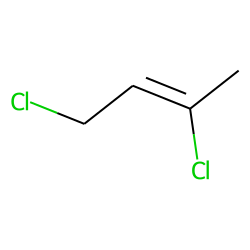
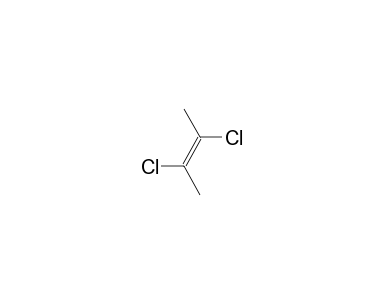
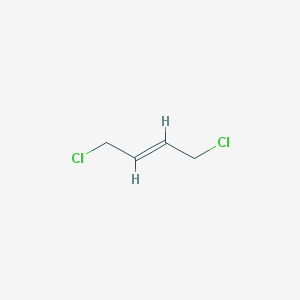
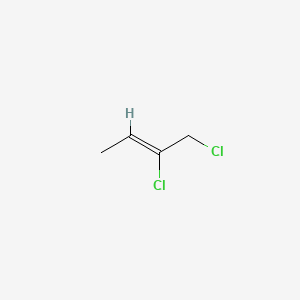
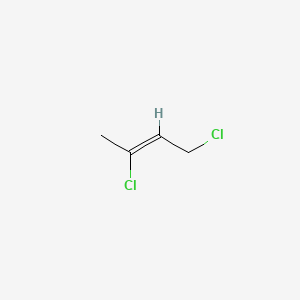
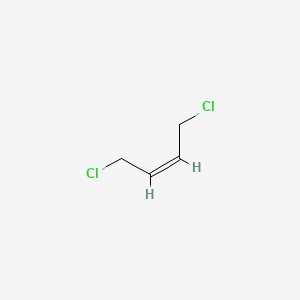
2Z)-1,3-dichloro-2-butene - 926-57-8, C4H6Cl2, density, melting point, boiling point, structural formula, synthesis

Indicate whether each of the following molecules is capable of geometrical (cis-trans) isomerism. a. 2,3-dichlorobutane b. 2,3-dichloro-2-butene c. 1,3-dimethylbenzene d. 4,4-dimethyl-2-pentyne | Homework.Study.com

![1,3-Dichloro-2-butene - Optional[1H NMR] - Chemical Shifts - SpectraBase 1,3-Dichloro-2-butene - Optional[1H NMR] - Chemical Shifts - SpectraBase](https://spectrabase.com/api/spectrum/FExxRL1uIuI/structure.png?h=300&w=382)